| Honors Chemistry - Chemical Bonding
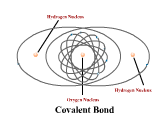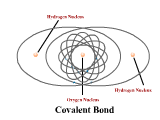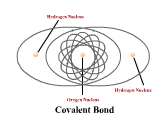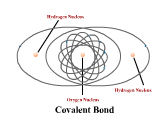
1 - Explain how atoms combine to form compounds through both ionic and covalent bonding. 2 - Explain why chemical bonds occur.
3 - Use electronegativity to explain the difference among ionic, polar, and nonpolar covalent bonds. (4.3)
4 - Draw Lewis dot structures for simple ionic and covalent compounds (including polyatomic ions). (4.2)
5 - Predict chemical formulas based on the number of valence electrons. (4.1)
6 - Describe the process of metallic bond formation.
7 - Use valence-shell electron-pair repulsion theory (VSEPR) to predict the molecular geometry (linear, bent, triangular planar, triangular pyramidal and tetrahedral) of simple molecules. (4.4)
8 - Identify how hydrogen bonding in water affects a variety of physical, chemical, and biological phenomena (e.g., surface tension, capillary action, density, boiling point). (4.5)
|
|


